Artificial Photosynthetic Assemblies | Spin Effects in Solar Energy Harvesting | Metal Organic Frameworks for Chemical Warfare Agent Degradation
Artificial Photosynthetic Assemblies

Natural photosynthetic systems utilize the sun’s energy to transform carbon dioxide and water into carbohydrates, nature’s stored solar fuel. Artificial photosynthetic assemblies need to be able to absorb sunlight, transfer absorbed energy, transport charge, oxidize water and reduce carbon dioxide to a fuel source. Metal-Organic Frameworks (MOFs) can be functionalized to provide an efficient, heterogeneous system for artificial photosynthesis.
Spin Effects in Solar Energy Harvesting
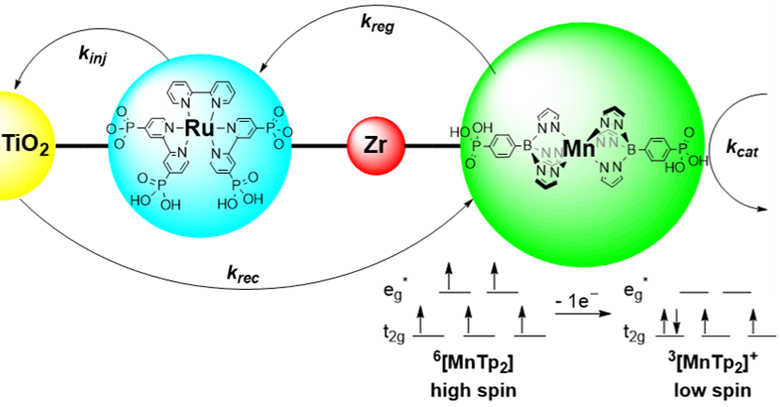
Long-lived, charge-separated states are critical to the realization of both photovoltaic and photocatalytic assemblies. The Morris group recently demonstrated the ability to control the lifetime of photo-induced charge separated states by employing a charge-transfer-induced, spin-transition (high-spin to low-spin transition) in a class of manganese poly(pyrazolyl)borates. Our current research effort is motivated by the need to understand the role of molecular reorganization and metallic spin state in controlling charge-separated-state lifetimes and catalytic selectivity/efficiency.
Metal Organic Frameworks for Chemical Warfare Agent Degradation
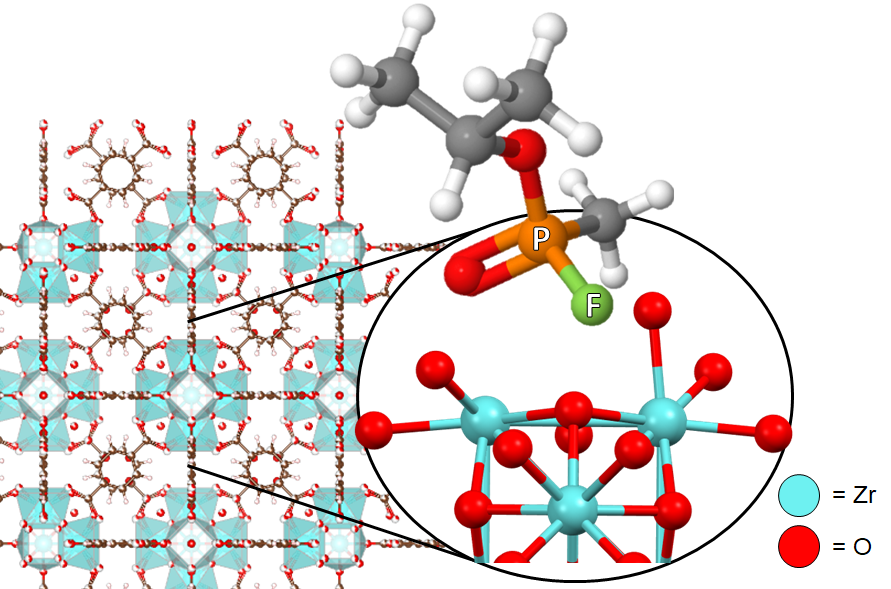
Zirconium (and Hafnium) based metal-organic frameworks (MOFs) have shown immense promise in the sorption and solution-phase degradation of chemical warfare agents (CWAs). Their activity has been attributed to open coordination sites on the MOF nodes and the porous structure of MOFs which enables rapid diffusion to these sites. That said, recent studies show that only 30% of the MOF’s surface area is utilized during sorption and reactivity. Additionally, the products of CWA hydrolysis poison the catalysts during gas-phase reactivity. The Morris group aims to address these challenges through synthetic manipulation of MOF structure and active-site engineering. Thw work is highly collaborative with joint efforts in the John Morris group (VT, Chemistry), Ayman Karim group (VT, Chemical Engineering), Diego Troya group (VT, Chemistry), Yue Wu group (UNC, Physics), Karl Johnson group (Pitt, Chemical Engineering), and the Chemical and Biological Center in Edgewood, MD.



